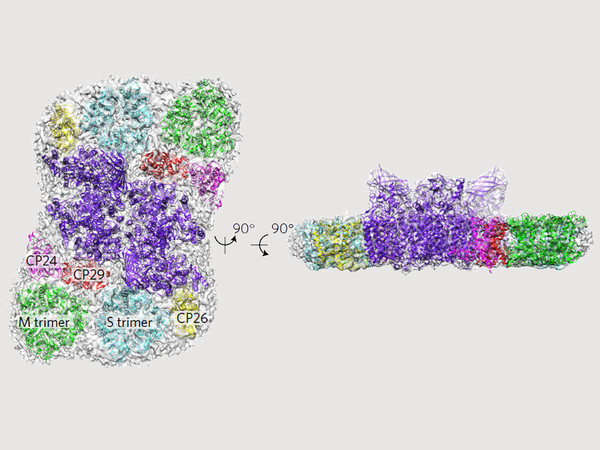
The first three-dimensional model of the largest stable photosystem II supercomplex, in never-before-seen detail, is the product of international research in which Roman Kouřil and Ravindra S. Kale, representing Olomouc’s Centre of the Haná Region for Biotechnology and Agriculture took part, in addition to scientists from the Netherlands and France. The result of their roughly two-year work was recently published in the prestigious scientific journal Nature Plants. Knowledge of the structure of photosystem II is the key to not only understanding the method of transferring absorbed sunlight and its use in photosynthesis, but also for example in understanding acclimation strategies of plants under changing light conditions.
In addition to transmission electron microscopy, scientists also used cryo-electron microscopy in the study – for which its developers were awarded the 2017 Nobel Prize in Chemistry. It was this technological development in the field which allowed the scientists to study large protein supercomplexes in 3D, at atomic resolutions. The undisputed advantage of high-resolution cryo-electron microscopy is that the observed protein complex does not have to be in crystal form, as in conventional X-ray crystallography structural analyses.
“Our work is the first study of the largest photosystem II supercomplexes in 3D at such high resolution. We based it on our research from 2009, when we utilised the classic method of electron microscopy to obtain a 2D structure, with a resolution of 12 Ångströms, i.e. 0.12 nanometres. But we had ambitions to take it further. Thanks to the improved method of cryo-electron microscopy, we have now achieved a 3D resolution of 5.3 Ångströms, which was a huge surprise, even to us,” said biophysicist Roman Kouřil.
According to him, research into this area in recent years has attracted more teams throughout the world, attempting to push the limits even further. The huge “boom” in cryo-electron microscopy occurred after the introduction of the new type of electron detectors in 2012–2013, thanks to which detection is much faster and more sensitive.
The photosystem II supercomplex is composed of a core, whose structure is similar in all photosynthetic organisms. In plants, this core is bound by a number of specific antenna complexes which absorb light. “The question is, why do plants have such an arrangement of the light-harvesting antenna complex, and what roles its individual components have? Thanks to basic research, we are gaining information about which way the absorbed light energy is transferred to the core and the reaction centre of the photosystem. Detailed knowledge of the organisation of pigments in the antenna complexes is very important for understanding how the photosystem is able to properly use light energy, or – if there is a surplus – how it is able to safely get rid of it,” explained Kouřil. Together with his colleagues, he also succeeded in clarifying the specific qualities of the supercomplex, especially the flexibility of the supercomplex’s antenna photosystem.
The experimental work was carried out mostly in the Netherlands at the national NeCEN microscopy centre at Leiden University and at the University of Groningen, where Roman Kouřil worked as a postdoc for nine years. Even after returning to Palacký University Olomouc, he kept in touch with his former colleagues. For example, three years ago, they jointly carried out a structural characterisation of another protein supercomplex, photosystem I – NAD(P)H dehydrogenase, which is one of the important functional units of plants’ photosynthetic apparatus.